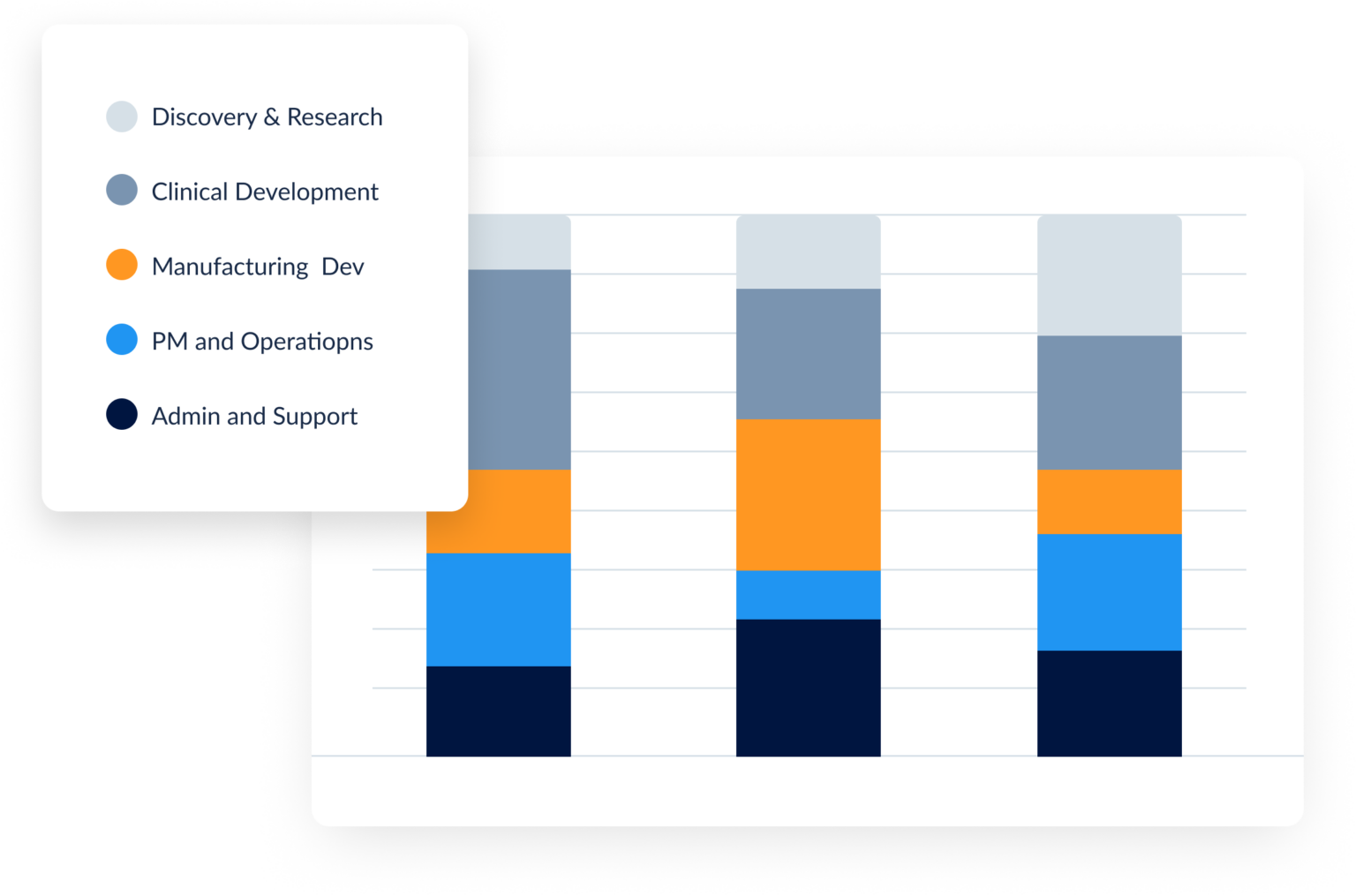
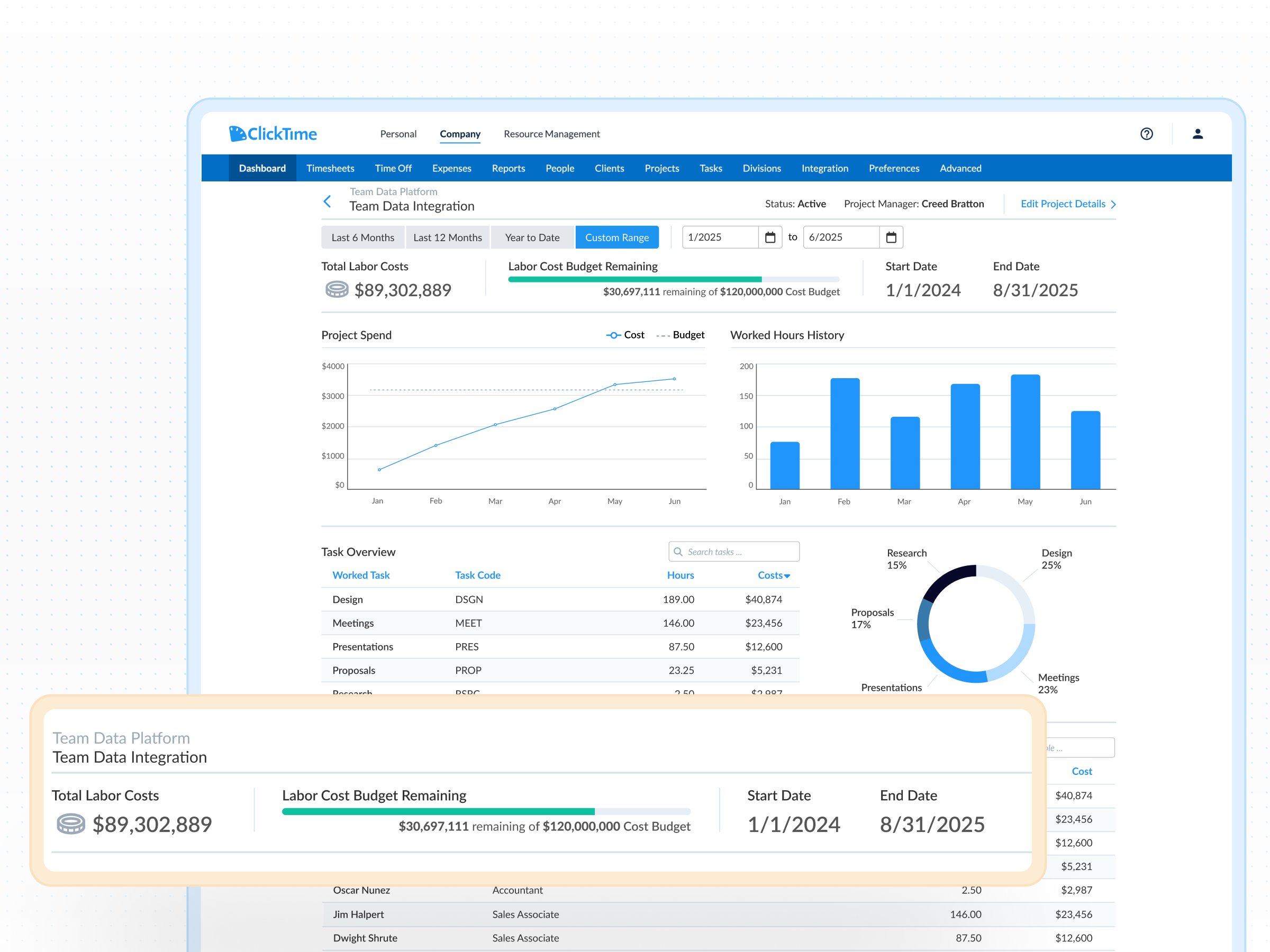
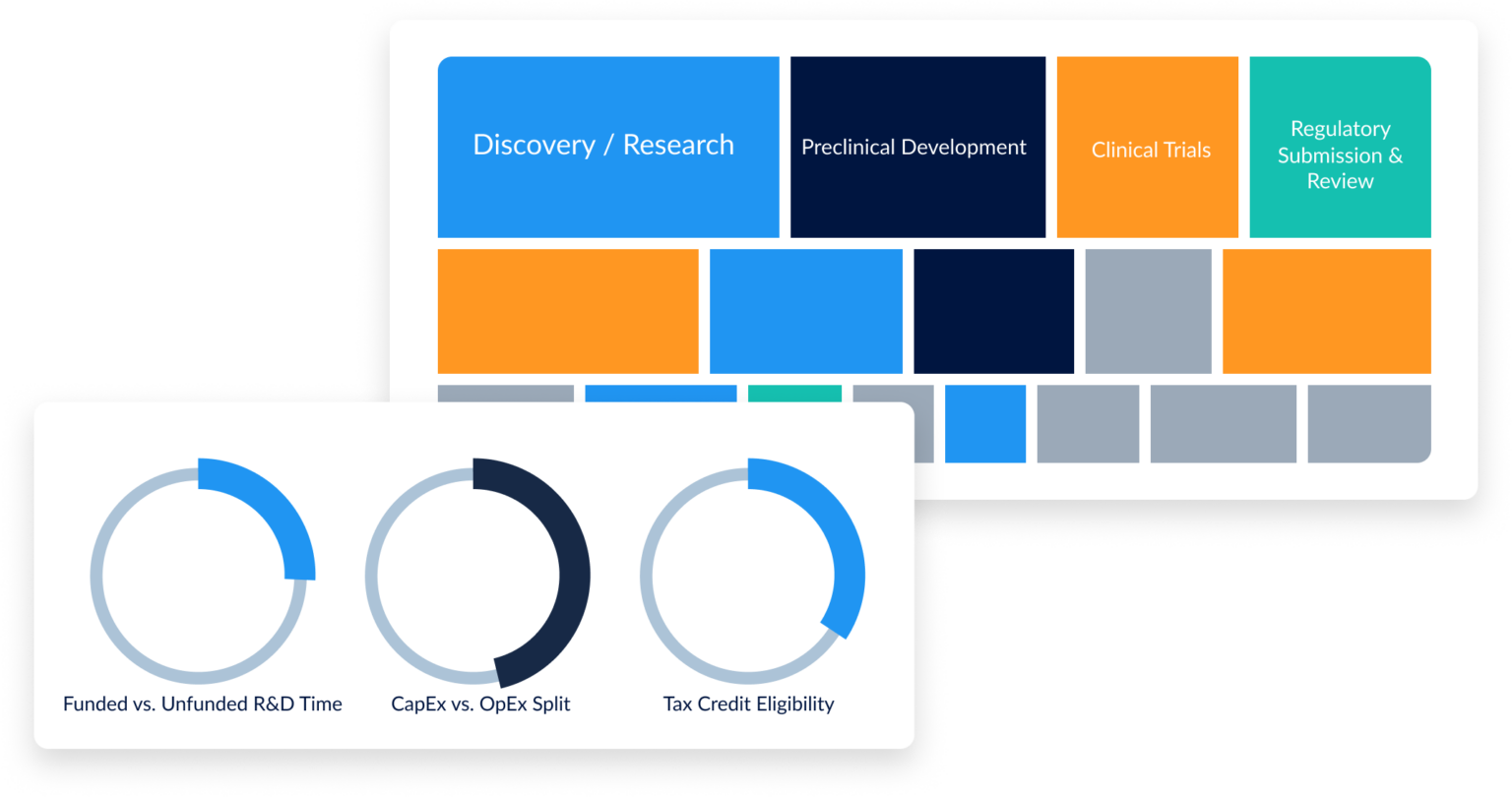
Qualified research activities under §41 must meet a four-part test: the work must be technological in nature, intended to develop a new or improved business component, involve a process of experimentation, and address technical uncertainty. For biotech companies, this commonly includes drug discovery, clinical trial design, assay development, process optimization, and formulation work. The key challenge is documenting which hours were spent on qualifying vs. non-qualifying activities—something project-level time tracking solves at the point of entry.